Translation from Bench to Bedside and back
In focusing on the translation of basic research discoveries to clinical testing and back, our URPP aims at assessing and improving the beneficial effects of novel single or combined treatment strategies and simultaneously minimizing the adverse effects.
The main aims of this part can be summarized as follows:
- Understanding and avoiding treatment-associated adverse effects
- Developing improved treatments for cancer
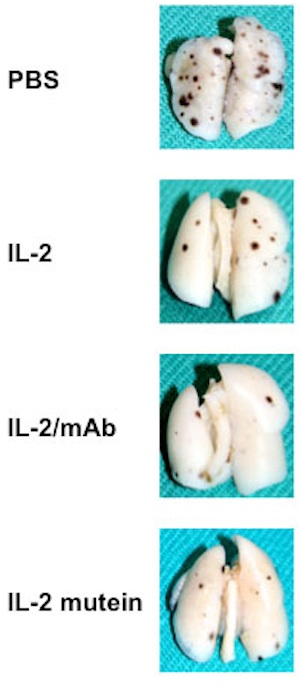
Ad 1. Currently, most studies examining the efficacy of a novel drug in an animal model fail to assess the potential adverse side effects of the drug. Yet, many treatments, including immunotherapy, often affect different organs, such as the lungs, liver, and kidneys, even if such therapeutics appear to be well-tolerated by the individual. The screening for adverse side effects of novel immunotherapeutics already at an early stage of drug development will help us identify well-tolerated from more toxic approaches. Moreover, these efforts will extend our knowledge in this field, which is becoming increasingly important for academic and pharmaceutical drug development by helping to better select candidate therapeutics for costly clinical trials.
Ad 2. The faithful generation and further testing of promising cancer treatments is a crucial step towards clinical studies, which will be a central part of this URPP with focus on translational cancer research. During the first periods of this URPP grant, we have generated promising therapeutic approaches, which we intend to test in investigator-initiated clinical trials, pending additional funding by other sources.
The three examples listed below illustrate a therapeutic strategy that will be further developed and tested within this URPP.
a) IL-2/anti-IL-2 mAb complexes
b) Use of IL-12 fusion protein in combination with anti-CTLA-4 mAb
c) Use of any of the following: anti-HSC/AML CAR T cells (Lentiviral vector or mRNA based) or bi- or multi-specific T-cell engager (BiTE) antibodies
Generation and testing of GMP-grade novel therapeutics (such as human IL-2/anti-human IL-2 mAbs and huIL-12-Fc, CAR T-cells, bispecific antibodies) will place us in an ideal position to find sufficient external financing for proceeding to investigator-initiated clinical trials.